DFG Priority Program (SPP) 2240 eBiotech
DFG Priority Program (SPP) 2240 eBiotech
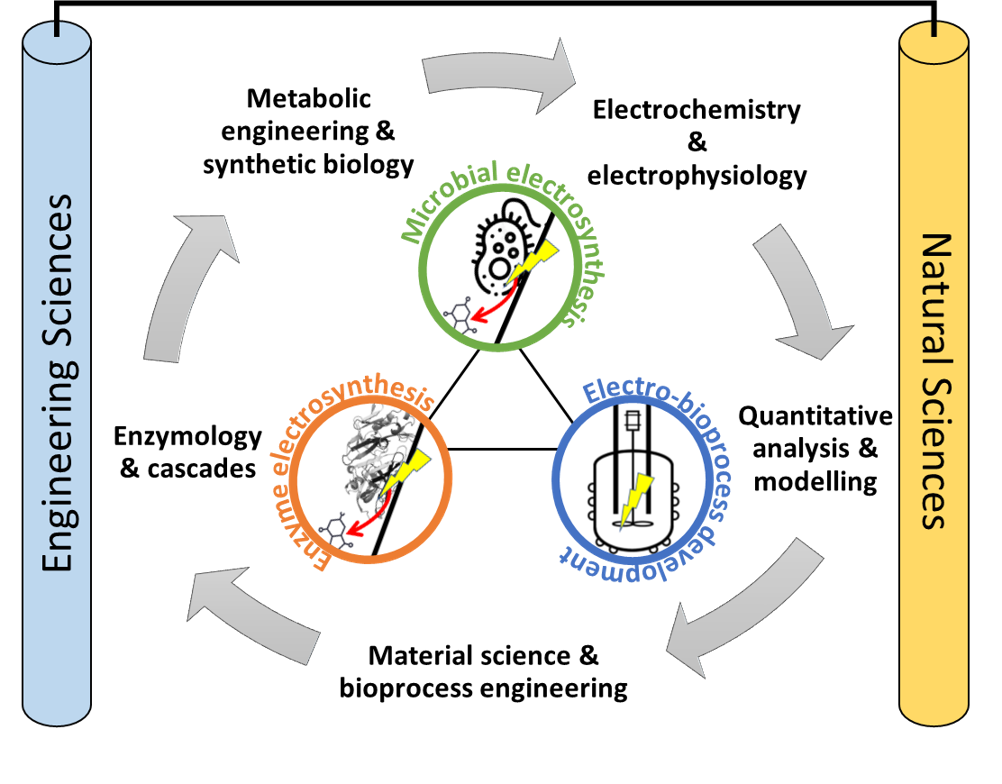
The priority program “Bioelectrochemical and Engineering Fundamentals to Establish Electro-biotechnology for Biosynthesis – Power to Value-added Products (eBiotech)” (SPP 2240) is located at the interface between natural sciences and engineering. Starting in 2021, the first phase of the DFG priority program eBiotech combines the competencies from 28 German research groups of these disciplines working in 14 projects, while the second phase continues with 21 German research groups working on 11 projects. Research is done in the form of a collaborative, interdisciplinary research network, with the second phase providing continuity to the first one. The focus of the DFG priority program 2240 eBiotech lies on the targeted development and efficient design of electro-biotechnological processes through a better understanding of the underlying processes.
The Priority Program 2240 eBiotech in a broader context
The Priority Program 2240 eBiotech in a broader context
The SPP 2240 “e-Biotech” harnesses the latest advances in (bio)electrochemistry, microbiology, materials science, systems and synthetic biology and process engineering to pave the way for an emerging new field, the electro-biotechnology. It provides an immense chance to develop completely new and highly efficient bioprocesses from electricity and sustainable substrates including CO2. To this end, fundamental research, especially with an engineering approach to link the basic science with process technology, are urgently needed. The SPP timely brings together research groups with broad expertise and from different locations for a true interdisciplinary collaboration. With this e-Biotech lays the necessary foundations for realizing the promise of electro-biotechnology and enabling Germany to become internationally leading in this important area of bioeconomy and the use of regenerative energy (Power-to-Products).